CTMX: Hard work meets frustration
Disclosures: This Report is provided by Fourier Transform Research solely for educational purposes and constitutes impersonal, generic commentary based entirely on publicly available data. The views expressed herein reflect the personal opinions of the author as of the date of publication; they are not tailored to any individual’s financial situation or objectives and do not constitute investment advice, a recommendation, or an offer to buy or sell any security. The author holds no position in the security discussed. No part of the author’s compensation is, was, or will be directly or indirectly related to any specific recommendation or view contained herein. The author has not received—and will not receive—any payment of any kind from the company(ies) whose securities are covered by this Report. This Report contains no material non‑public information. Trading the securities covered herein is subject to a blackout period for the author commencing 72 hours before and ending 24 hours after the Report’s public release. All factual data are believed to be accurate at the time of writing, but Fourier Transform Research makes no warranty as to its completeness or accuracy; recipients should conduct their own due diligence. Past performance is not indicative of future results.
1. EXECUTIVE SUMMARY
CX-2051 is a first-in-class, conditionally activated antibody-drug conjugate (ADC) targeting epithelial cell adhesion molecule (EpCAM) using CytomX’s proprietary Probody masking technology. The molecule is armed with CAMP59, a novel topoisomerase-1 inhibitor payload, at a drug-antibody ratio (DAR) of 8. Interim Phase 1 data (April 7, 2025 cutoff) demonstrated a 28% confirmed objective response rate (ORR) and 5.8-month median progression-free survival (PFS) in heavily pretreated, unselected 4th/5th-line metastatic colorectal cancer (mCRC) patients [1]. This substantially exceeds the efficacy of approved agents in this setting: fruquintinib achieved a 1.5% ORR and 3.7-month PFS in FRESCO-2 [2], and regorafenib achieved a 1.0% ORR and 1.9-month PFS in CORRECT [3].
The critical finding from a platform validation standpoint is the complete absence of pancreatitis and hepatotoxicity in the safety profile. EpCAM is widely expressed in normal epithelial tissues including the pancreatic ductal epithelium, bile ducts, and liver. Every prior attempt to systemically target EpCAM has produced dose-limiting pancreatitis, hepatotoxicity, or both [4][5][6][7]. The absence of these signals in CX-2051 patients provides the strongest clinical evidence to date that protease-dependent Probody masking can effectively shield normal tissues from ADC-mediated cytotoxicity while maintaining tumor-selective activation.
The dominant safety signal—diarrhea occurring in 78.3% of patients at therapeutic doses, with 21.7% grade 3 [1]—is consistent with a payload-mediated enterohepatic recycling mechanism rather than on-target, off-tumor EpCAM engagement. This distinction is critical to the investment thesis: the GI toxicity likely arises from biliary excretion and intestinal reactivation of free CAMP59 payload, a pharmacologic class effect shared by all camptothecin-derived ADCs. It is not evidence of Probody mask failure. This safety profile is manageable in the context of 4th-line mCRC where the current standard of care offers minimal clinical benefit.
Primary risks include the emerging colitis signal (1 case in 23 patients at therapeutic doses), the possibility that prophylactic loperamide may be insufficient to reduce grade 3 diarrhea to commercially acceptable levels, and the Q1 2026 expansion data readout representing a binary catalyst event. We do not recommend establishing positions ahead of the data readout given the high remaining uncertainty, but view the post-data risk/reward favorably if the expansion cohort confirms the Phase 1a findings.
Key Metrics Snapshot
2. THE EpCAM TARGET: BIOLOGY AND HISTORICAL FAILURES
2.1 EpCAM Biology and Expression
Epithelial cell adhesion molecule (EpCAM, also known as CD326 or TROP-1) is a transmembrane glycoprotein expressed on the basolateral surface of most epithelial tissues. In colorectal cancer, EpCAM expression is notably high and homogeneous, with immunohistochemistry (IHC) H-scores frequently exceeding 280 (maximum 300) [9]. This makes it an attractive ADC target from an antigen density perspective—sufficient for efficient receptor-mediated internalization and payload delivery.
However, EpCAM is not tumor-restricted. It is constitutively expressed on normal intestinal epithelium, pancreatic ductal cells, bile duct epithelium, hepatocytes, renal tubular cells, and pulmonary epithelium [10]. This broad normal-tissue expression has historically made systemic EpCAM targeting untenable, as therapeutic antibodies bind normal tissues with sufficient avidity to cause clinically meaningful organ damage.
2.2 History of EpCAM-Targeting Failures
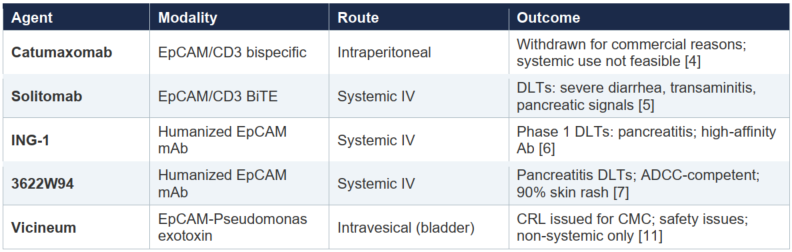
Multiple attempts to therapeutically target EpCAM have failed due to on-target, off-tumor toxicity:
The consistent pattern across these agents—pancreatitis, hepatotoxicity, severe GI toxicity—reflects the fundamental challenge of systemic EpCAM engagement. This history is what makes the CX-2051 safety data so remarkable.
3. CX-2051: MOLECULAR CHARACTERIZATION
3.1 Antibody Component
The antibody is a humanized IgG4 derived from a mouse anti-EpCAM antibody designated mEpCAM23. Humanization used human germline sequences IGKV2D-29*02 and IGHV1-3*01 as acceptor frameworks, producing the variant huEpCAM23Gv4.2 [12]. The IgG4 backbone was selected to minimize Fc-mediated effector functions (ADCC, CDC), reducing off-target immune activation—a deliberate design choice given the broad normal-tissue expression of EpCAM.
3.2 CAMP59 Payload
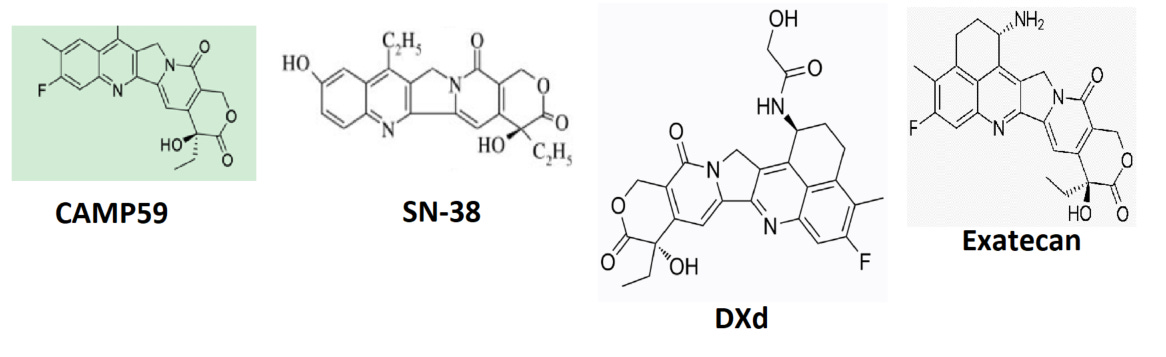
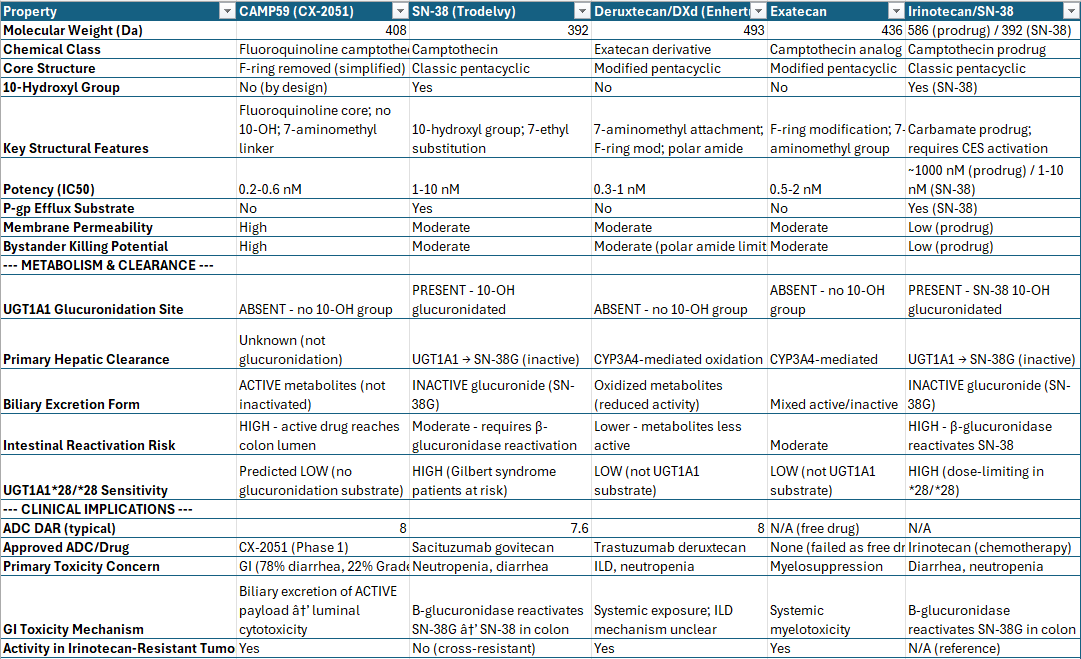
CX-2051 carries CAMP59, a novel camptothecin derivative discovered in collaboration with ImmunoGen (now AbbVie). CAMP59 has several structural features distinguishing it from existing Topo-1 payloads:
Efflux Resistance
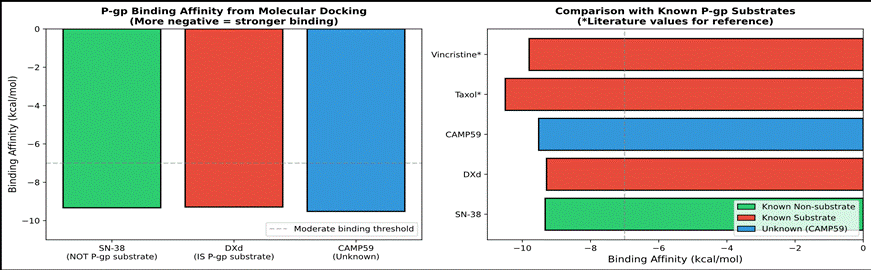
A key structural advantage of CAMP59 over SN-38 (the active metabolite of Trodelvy/sacituzumab govitecan) relates to drug efflux. A systematic screen of 27 common ADC payloads demonstrated that camptothecin itself was not a P-glycoprotein (P-gp) substrate, while SN-38 was identified as a substrate [14]. CAMP59 is structurally closer to the exatecan class—it lacks the 10-hydroxyl group present in SN-38 and carries an 11-fluoro substitution. This is consistent with the camptothecin structural subclass found to be resistant to P-gp efflux. While no specific Caco-2 efflux data has been published for CAMP59, its structure falls within the efflux-resistant camptothecin subclass. Molecular docking simulations with P-gp, BCRP, and MRP1 transporters support this assessment, though the in silico data alone is insufficient to definitively classify efflux liability.
Figure 1: P-gp binding affinity from molecular docking. CAMP59 binding falls between SN-38 and DXd, consistent with intermediate-to-low efflux substrate potential. Docking data should be interpreted cautiously.
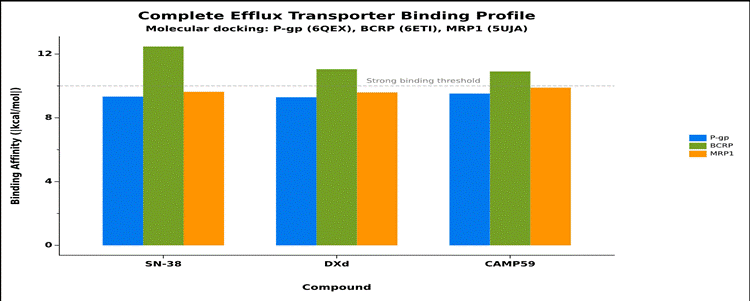
Figure 2: Complete efflux transporter binding profile across P-gp, BCRP, and MRP1. CAMP59 shows broadly comparable binding across transporters, with no outlier signals suggesting strong substrate activity.
4. PROBODY MASKING TECHNOLOGY: MECHANISM AND CLINICAL VALIDATION
4.1 Mechanism of Conditional Activation
The Probody platform addresses the EpCAM targeting challenge by incorporating a proprietary masking peptide on the antibody Fab arms that physically blocks antigen binding in normal tissues. The mask is tethered to the antibody via a protease-cleavable linker. In the tumor microenvironment, where protease activity is substantially elevated, the linker is cleaved by tumor-associated proteases including matriptase, urokinase, MMP-2, MMP-9, and legumain, releasing the mask and restoring full antibody binding capacity [15].
In healthy tissues—even those expressing EpCAM at high levels such as intestinal epithelium, bile ducts, and pancreatic ducts—protease activity is much lower than in tumor tissue. In situ zymography (IHZ) analysis demonstrated that CRC, lung, and other tumor tissues had activating protease activity while normal tissues lacked it [15]. When masked, CX-2051 exhibits approximately 70-fold reduced binding affinity and approximately 30-fold reduced cytotoxicity compared to the unmasked form [1].
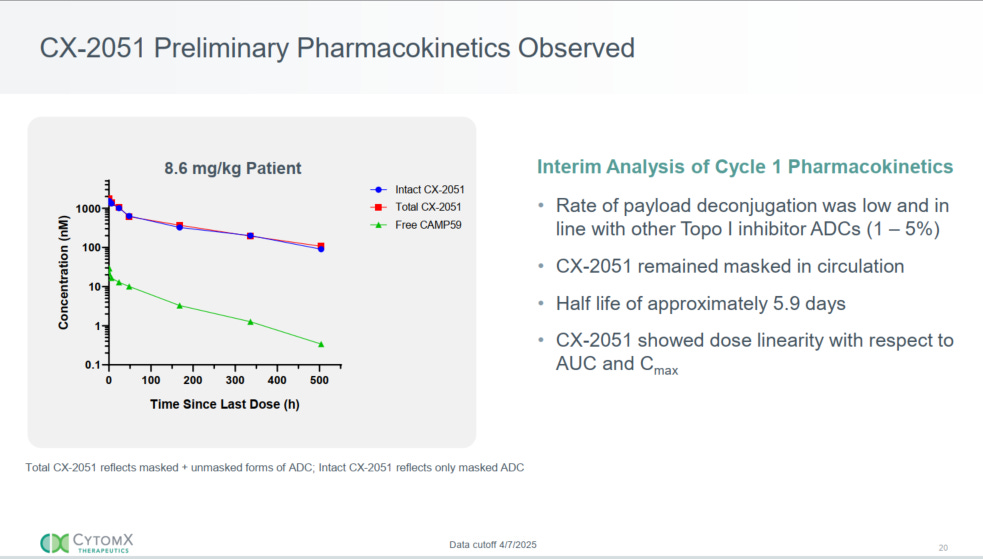
4.2 Pharmacokinetic Validation of Masking
A critical piece of platform validation emerged from the May 2025 data presentation. CytomX showed that the intact (masked) versus total antibody PK profiles were essentially identical in patients [1][16]. This means that very little premature unmasking is occurring in systemic circulation or normal tissues. If significant off-target cleavage were happening, the masked species would deplete faster than total antibody. The overlapping PK profiles provide direct pharmacokinetic evidence that:
1. The mask remains intact during systemic circulation
2. Circulating proteases are not prematurely activating the Probody at clinically meaningful rates
3. Target-mediated drug disposition (EpCAM binding in normal tissues) is effectively suppressed by the mask
4.3 Clinical Safety Validation
The most compelling validation of the Probody platform is the clinical safety profile itself. Zero pancreatitis and minimal hepatotoxicity signals have been observed across all dose levels evaluated [1]. Given that every prior systemic EpCAM-targeting agent produced dose-limiting pancreatitis or hepatotoxicity [4][5][6][7], the absence of these signals in CX-2051 patients cannot be attributed to chance. It reflects functional masking preventing antibody engagement with EpCAM-expressing cells in the pancreas and liver. The alanine aminotransferase (ALT) and aspartate aminotransferase (AST) elevations observed (8.7% each, all grade 1-2) were within the range expected for patients receiving cytotoxic therapy and do not suggest direct hepatocellular injury [1].
Concerns remain regarding potential MMP-14 and MMP-9 expression in cancer patients outside the tumor microenvironment, which could theoretically lead to some degree of on-target, off-tumor mask cleavage [16]. However, the clinical PK and safety data to date suggest this is not a significant contributor.
5. PHASE 1 CLINICAL DATA
5.1 The Efficacy Bar in 4th-Line mCRC
It is important to contextualize CX-2051’s results against the current standard of care. In 4th-line and later mCRC, therapeutic options are extremely limited. Approved agents in this setting achieve low single-digit ORRs:
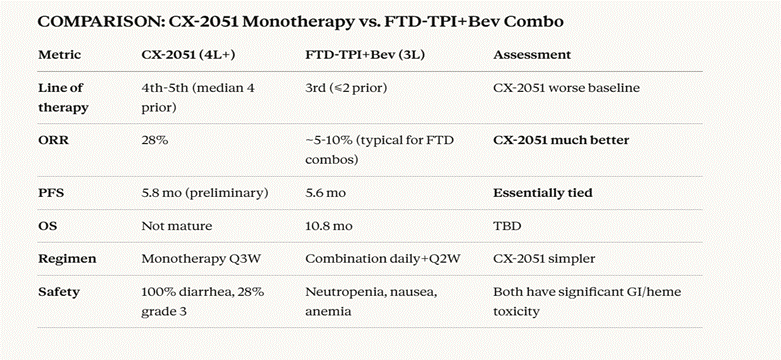
Figure 3: Head-to-head comparison of CX-2051 monotherapy versus the FTD-TPI+bevacizumab combination regimen (SUNLIGHT trial). Despite treating more heavily pretreated patients in a worse baseline setting, CX-2051 demonstrates substantially higher ORR with a simpler monotherapy regimen.
5.2 Efficacy Results
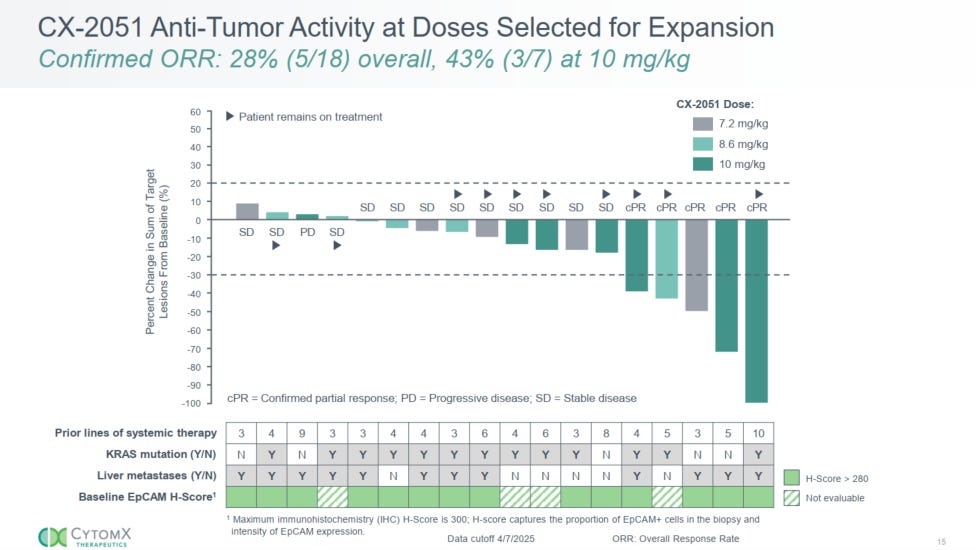
As of the April 7, 2025 data cutoff, 25 advanced mCRC patients were treated across dose levels 1-5. Eighteen patients at the three expansion dose levels (7.2, 8.6, and 10 mg/kg Q3W) were efficacy-evaluable [1]:
• Confirmed ORR: 28% (5/18) by RECIST v1.1
• Disease control rate (DCR): 94%
• Median PFS: 5.8 months
• ORR at 10 mg/kg: 43% (3/7 evaluable patients)
• 10/18 patients remained on treatment at data cutoff
These patients had a median of 4 prior lines of therapy. All had previously received irinotecan. Sixty-four percent had liver metastases, 64% harbored KRAS mutations, and 96% were microsatellite stable (MSS). Patients were not preselected based on EpCAM expression [1]. The inclusion of a complete response in a 10th-line patient is particularly notable given the depth of prior treatment.
We do not expect the ORR to meaningfully improve with data maturation. ADC-mediated tumor shrinkage typically manifests within 2-3 cycles, and the initial assessment window has passed for most enrolled patients. The PFS may extend modestly in higher dose cohorts if patients tolerate sufficient cycles. A PFS of 6 months or greater at 10 mg/kg would be a strong signal, though patients must maintain adequate performance status through enough cycles to achieve this.
6. SAFETY ANALYSIS
6.1 Adverse Event Profile
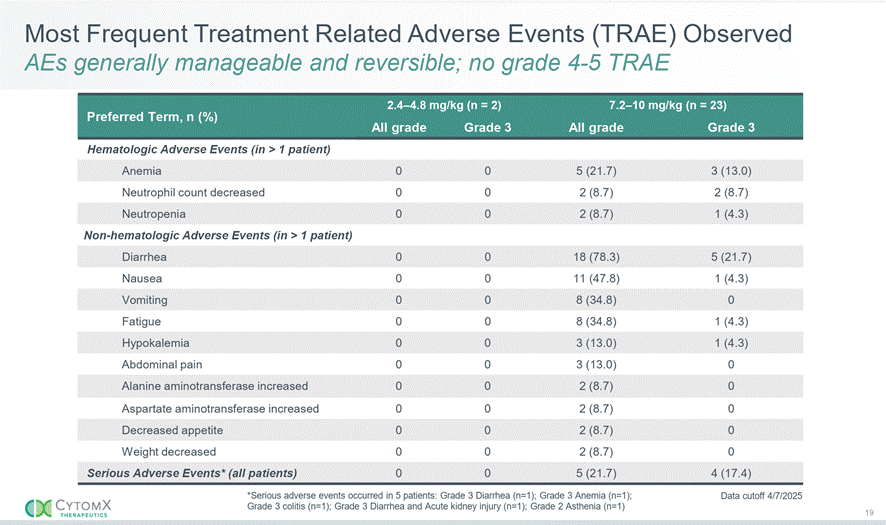
Figure 4: Most frequent treatment-related adverse events (TRAE) observed in the CX-2051 Phase 1 study. Data cutoff April 7, 2025. No grade 4-5 TRAE. Source: CytomX Therapeutics [1].
The safety profile is dominated by gastrointestinal and hematologic toxicities at therapeutic doses (7.2-10 mg/kg). Key observations:
• Diarrhea: 78.3% all-grade, 21.7% grade 3 (n=5 of 23 patients at therapeutic doses)
• Nausea: 47.8% all-grade, 4.3% grade 3
• Anemia: 21.7% all-grade, 13.0% grade 3
• Pancreatitis: 0% — critically absent
• Hepatotoxicity: ALT/AST elevations 8.7%, all grade 1-2; no drug-induced liver injury pattern
• Serious AEs: 21.7% (5 patients): G3 diarrhea (n=5), G3 anemia (n=1), G3 colitis (n=1), G3 diarrhea + AKI (n=1), G2 asthenia (n=1) [1]
• Grade 5 event: 1 treatment-related AKI in a patient with a solitary kidney (reported August 2025) [17]
6.2 Mechanistic Interpretation of GI Toxicity
The diarrhea signal warrants detailed mechanistic discussion because understanding its origin is central to evaluating the Probody platform and the drug’s commercial viability.
Enterohepatic Recycling and Payload Reactivation
Camptothecin-class payloads undergo a well-characterized enterohepatic recycling pathway [18][19]:
1. Free payload is released from the ADC after lysosomal processing in tumor cells.
2. Systemically circulating payload undergoes hepatic glucuronidation (if it carries a phenolic hydroxyl or aromatic amine) to form an inactive conjugate.
3. The glucuronide conjugate is excreted into bile and enters the intestinal lumen.
4. Bacterial β-glucuronidase in the gut cleaves the glucuronide, regenerating the active cytotoxic payload [18].
5. Reactivated payload damages intestinal epithelium, triggering secretory diarrhea [19][20].
The CAMP59 Structural Distinction
However, CAMP59 differs structurally from SN-38 and DXd in a critical way. CAMP59 lacks the 10-hydroxyl group that serves as the primary substrate for UGT-mediated glucuronidation. In its place, CAMP59 carries a fluorine at the corresponding position. This means the classical glucuronidation-based inactivation pathway likely does not apply to CAMP59—there is no obvious inactivation mechanism for the free payload. Consequently, the full amount of CAMP59 excreted into the GI tract via biliary clearance arrives in an active state without requiring bacterial reactivation.
This has important implications. Because CAMP59 was specifically designed for enhanced membrane permeability and potent bystander killing [13], the payload arriving in the intestinal lumen in its fully active form would be expected to produce direct epithelial cytotoxicity. This likely explains both the high rate of diarrhea and the observed colitis signal. Importantly, this mechanism is entirely independent of EpCAM engagement—it is a function of payload pharmacology and biliary excretion.
UGT1A1 Polymorphism Considerations
The UGT1A1*28 polymorphism, which reduces glucuronidation capacity, is a well-established risk factor for severe diarrhea with SN-38-containing regimens. In the general population, approximately 44% are heterozygous (*1/*28) and 11% are homozygous (*28/*28) [21]. However, since CAMP59 may not rely on glucuronidation for inactivation, the UGT1A1 genotype may be less relevant to CX-2051 diarrhea than it is for Trodelvy or irinotecan-based therapies. This requires further investigation.
6.3 Prophylactic Loperamide Assessment
CytomX has implemented prophylactic loperamide in the expansion cohorts. Loperamide acts on mu-opioid receptors in the intestinal myenteric plexus, reducing motility and increasing water reabsorption [22]. While this addresses the motility component of diarrhea, it does not address the direct epithelial cytotoxicity from active CAMP59 in the intestinal lumen. If the primary mechanism is payload-mediated mucosal injury rather than secretory hypermotility, loperamide may have limited efficacy in reducing grade 3 events. For some patients, slowing intestinal transit may paradoxically increase exposure time of active CAMP59 to the mucosa. Rescue octreotide remains available for refractory cases.
6.4 Colitis Risk: A Major Watch Item
One grade 3 colitis event has been reported in 23 patients at therapeutic doses (4.3%) [1]. While a single case is insufficient to establish a reliable incidence rate, this signal requires careful monitoring because:
• The mechanism—direct mucosal cytotoxicity from active payload—is plausible and potentially dose-dependent.
• Colitis carries substantially higher morbidity than diarrhea, with risks including perforation, surgery, and fatality.
• Sacituzumab govitecan (Trodelvy), which carries the SN-38 payload, has an approved label colitis rate of approximately 2% (serious AE) [23]. If CX-2051 colitis rates exceed 5-10%, this could become a significant label issue.
• Rare colitis cases are a known class effect in irinotecan regimens, and ipilimumab-associated colitis provides a reference point for immune-mediated GI inflammation management [24].
This is the single largest risk factor for the program beyond the binary data readout. If the expansion cohort colitis rate increases to >5%, the safety narrative becomes considerably more complicated.
7. INVESTMENT SCENARIOS AND MARKET REACTION
7.1 Q1 2026 Data Readout Framework
Figure 5: Anticipated market reaction scenarios for the Q1 2026 data readout. The three-variable framework (ORR, PFS, grade 3 diarrhea rate) captures the key dimensions of risk.
The Q1 2026 expansion data update represents a binary catalyst event. With approximately 100 patients enrolled across three dose levels (7.2, 8.6, 10 mg/kg), the dataset will have substantially more statistical power to characterize both efficacy and safety. Key thresholds for market interpretation:
• Strong positive: ORR >28% + PFS >6 mo + Grade 3 diarrhea <20%. Colitis <5%. Confirms/exceeds May 2025 data.
• Modest positive: ORR 25-28% + PFS 5.5-6 mo + Grade 3 diarrhea 20-22%. Colitis 5-10%. Confirms May data with caveats.
• Negative: ORR <25% or PFS <5.5 mo or Grade 3 diarrhea >23%. Colitis >10%. Regression from May data.
7.2 Positioning Recommendation
We do not recommend establishing positions ahead of the binary Q1 2026 readout. The residual risk remains high: small patient numbers could produce regression in ORR, the colitis signal could worsen materially, or unexpected hepatotoxicity could emerge at expanded enrollment. However, buying after a confirmatory data readout will derisk the event substantially while still offering significant upside into Phase 2 and registrational development.
Figure 6: 4th-line monotherapy revenue model (US only). Peak US sales estimated at $936M based on 26,000 treatable patients, 45% market share, and $80,000 average revenue per patient course.
8. COMMERCIAL OPPORTUNITY
8.1 Epidemiology and Patient Flow
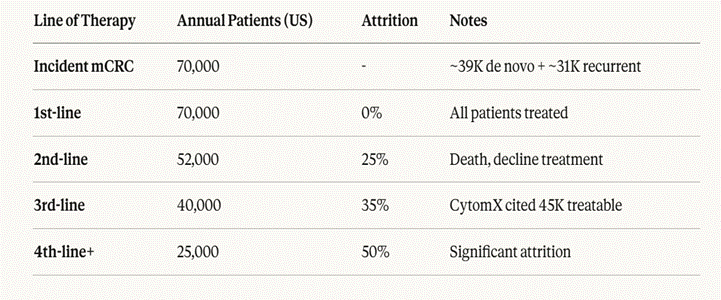
Figure 7: Annual US patient flow by line of therapy in metastatic CRC. Approximately 70,000 incident mCRC patients per year (~39,000 de novo + ~31,000 recurrent), with significant attrition at each successive line. The 4th-line addressable population is approximately 25,000 patients per year. Source: ACS, CytomX investor presentations [1][25].
8.2 Pricing Benchmarks
Expected CX-2051 pricing in the context of comparable oncology ADCs and late-line mCRC agents:
• Estimated dose cost: $38,000-42,000 per infusion (Q3W)
• Monthly equivalent: ~$52,000-58,000
• Estimated annual list price: $170,000-185,000
For reference, fruquintinib (Fruzaqla) carries an annual WAC of approximately $190,000-200,000. Enhertu (trastuzumab deruxtecan) lists at approximately $165,000-180,000 annually. CX-2051 pricing in this range would be competitive, particularly given the monotherapy convenience and superior ORR versus approved alternatives.
8.3 Revenue Modeling
The 4th-line monotherapy opportunity represents the near-term commercial case. At 26,000 annual treatable patients in the US, 45% market share at maturity, and $80,000 average revenue per patient treatment course, peak US sales are estimated at approximately $936M. Adding ex-US markets at a standard 60/40 US/ROW split suggests global peak sales of approximately $1.5B from the 4th-line monotherapy indication alone [25].
8.4 Expansion Opportunities
CytomX has announced plans to initiate a Phase 1b combination study with bevacizumab in CRC in Q1 2026 [17]. If the combination shows acceptable safety and PFS exceeding 8 months in earlier-line settings, the addressable patient population expands substantially (2nd/3rd-line mCRC comprises approximately 52,000-70,000 annual US patients). We note this as potential upside optionality but do not model it at this stage given the early development timeline.
9. SUMMARY AND CONCLUSIONS
CX-2051 represents a meaningful advance in two respects. First, it provides clinical proof-of-concept that protease-activated masking technology can unlock previously intractable targets. The absence of pancreatitis and hepatotoxicity in a setting where every prior EpCAM-targeting agent produced these DLTs is not incremental—it is a step-change in the feasibility of EpCAM-directed therapy. Second, the 28% ORR and 5.8-month PFS in unselected 4th/5th-line mCRC patients represent a genuine clinical advance over approved standards of care that achieve low single-digit ORRs.
The primary safety concern—GI toxicity including diarrhea and colitis—is mechanistically attributable to payload-mediated enterohepatic recycling and direct mucosal cytotoxicity, not to Probody mask failure or on-target/off-tumor EpCAM engagement. This distinction matters because it means: (a) the Probody platform is validated, and (b) the GI toxicity is a pharmacology problem, potentially addressable through dose optimization, supportive care refinement, or combination strategies that reduce payload exposure to the GI mucosa.
Key conclusions:
9. Platform validated: Zero pancreatitis, minimal liver signals, and overlapping masked/total PK profiles provide the strongest clinical evidence to date for the Probody conditional activation approach.
10. Efficacy exceeds SOC by wide margin: 28% ORR vs. 1-2% for approved agents in the same setting. This is commercially viable in 4th-line mCRC.
11. GI toxicity is payload-driven, not target-driven: Diarrhea reflects enterohepatic recycling of active CAMP59, a class effect that may be partially addressable.
12. Colitis is the key risk to monitor: A rate exceeding 5-10% would materially impact the commercial profile and regulatory path.
13. Approval path exists in 4th-line mCRC: Assuming expansion data confirms Phase 1a results and colitis does not escalate materially, accelerated approval on ORR is feasible.
14. High risk pre-data, favorable post-data: Do not establish positions ahead of the Q1 2026 readout. Confirmatory data substantially derisks the investment and the transition into Phase 2 still offers meaningful upside.
REFERENCES
[1] CytomX Therapeutics. Positive Interim Data From Phase 1 Dose Escalation Study of CX-2051 in Advanced CRC. Press Release, May 12, 2025. CytomX corporate slide deck, data cutoff April 7, 2025.
[2] Dasari A, Lonardi S, Garcia-Carbonero R, et al. Fruquintinib versus placebo in patients with refractory metastatic colorectal cancer (FRESCO-2). Lancet. 2023;402(10395):41-53. doi:10.1016/S0140-6736(23)00772-9
[3] Grothey A, Van Cutsem E, Sobrero A, et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):303-312.
[4] Sebastian M, Passlick B, Friccius-Quecke H, et al. Treatment of malignant pleural effusion with the trifunctional antibody catumaxomab (Removab): results of a phase 1/2 study. J Immunother. 2007;30(1):81-91.
[5] Kebenko M, Goebeler ME, Wolf M, et al. A multicenter phase 1 study of solitomab (MT110), a bispecific EpCAM/CD3 T-cell engager (BiTE) antibody construct, in patients with refractory solid tumors. Oncoimmunology. 2018;7(8):e1450710.
[6] de Bono JS, Tolcher AW, Forero A, et al. ING-1, a monoclonal antibody targeting EpCAM in patients with advanced adenocarcinomas. Clin Cancer Res. 2004;10(22):7555-7565.
[7] LoBuglio AF, Saleh MN, Lee J, et al. Phase I trial of 3622W94 anti-epithelial cell adhesion molecule monoclonal antibody in patients with advanced adenocarcinoma. Proc Am Soc Clin Oncol. 1997.
[8] CytomX Therapeutics. 2025 Strategic Pipeline Priorities and Corporate Update. Press Release, January 6, 2025.
[9] CytomX Therapeutics corporate presentation, May 2025. IHC H-score data for EpCAM in CRC tissues.
[10] Trzpis M, McLaughlin PM, de Leij LM, Harmsen MC. Epithelial cell adhesion molecule: more than a carcinoma marker and adhesion molecule. Am J Pathol. 2007;171(2):386-395.
[11] Sesen Bio. Vicineum (oportuzumab monatox) FDA regulatory history. CRL issued for CMC deficiencies, 2021.
[12] CytomX Therapeutics. CX-2051 preclinical characterization data. Patent filings and conference presentations.
[13] Kovtun YV, Audette CA, Mayo MF, et al. Antibody-maytansinoid conjugates designed to bypass multidrug resistance. Cancer Res. 2010;70(6):2528-2537. PMC6792168.
[14] Sauveur J, Matera EL, Naud C, et al. Identification of P-glycoprotein substrates among camptothecin ADC payloads: systematic screen of 27 compounds. PMC12154920. 2025.
[15] Desnoyers LR, Vasiljeva O, Richardson JH, et al. Tumor-specific activation of an EGFR-targeting Probody enhances therapeutic index. Sci Transl Med. 2013;5(207):207ra144. (Probody platform reference)
[16] CytomX Therapeutics. CX-2051 intact vs total antibody PK analysis. May 2025 investor presentation. Also: Nieri P, Dober B, Passalacqua R, et al. Role of tumor microenvironment proteases in Probody activation. Br J Cancer. 2017;114:233-240. doi:10.1038/bjc.2016.127
[17] CytomX Therapeutics. Update on CX-2051 Phase 1 Study. Press Release, August 13, 2025.
[18] Wallace BD, Wang H, Lane KT, et al. Alleviating cancer drug toxicity by inhibiting a bacterial enzyme. Science. 2010;330(6005):831-835. Also: Pharmacol Ther. 2022. doi:10.1016/S0009-2797(22)00151-X
[19] Stringer AM, Gibson RJ, Logan RM, et al. Irinotecan-induced mucositis manifesting as diarrhea corresponds with an amended intestinal flora and mucin profile. Int J Exp Pathol. 2009;90(5):489-499. Also: IJMS. 2020;21(14):4919. doi:10.3390/ijms21144919
[20] Curr Drug Metab. 2012;13(6):802-818. Review: enterohepatic recycling of camptothecin analogues.
[21] Wang Y, Shen L, Xu N, et al. UGT1A1 predicts outcome in colorectal cancer treated with irinotecan and fluorouracil. Cancer Cell Int. 2024. doi:10.1002/cac2.70058
[22] Baker DE. Loperamide: a pharmacological review. Rev Gastroenterol Disord. 2007;7(Suppl 3):S11-S18.
[23] TRODELVY (sacituzumab govitecan-hziy) Prescribing Information. Gilead Sciences, Inc. 2023. Colitis listed as serious adverse reaction in >1% of patients.
[24] Weber JS, Kahler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30(21):2691-2697.
[25] American Cancer Society. Colorectal Cancer Facts & Figures; CytomX Therapeutics corporate presentations 2025.
This report is for informational purposes only. It does not constitute investment advice, a recommendation to buy or sell securities, or an offer or solicitation of any kind. Past performance is not indicative of future results. All investment carries risk including loss of principal.